A doctor asked: “Why do I now see many children receiving the third MMR dose — especially those who have received the first dose at 9 months and the second dose at 12 months?”
Currently (since 2013), the American Advisory Committee on Immunization Practices (ACIP) recommends two doses of MMR for children, the first at 12 to 15 months, and the second before school entry, at 4 to 6 years.
The second dose can be given as early as four weeks (28 days) after the first dose and be counted as a valid dose if both doses were given after the child’s first birthday.
The second dose is not a booster, but is intended to produce immunity for the small number who fail to respond to the first dose. Two doses of the vaccine are recommended to ensure immunity and prevent outbreaks, as about 15 per cent of vaccinated children fail to develop immunity from the first dose.
The ACIP recommends the second dose to be administered upon entrance to kindergarten or first grade between the ages of about 4 and 6 years. This recommendation was based primarily on administrative considerations.
Childhood immunisation in the United States schedule diphtheria-tetanus-pertussis and poliovirus vaccinations before school entry. Simultaneous provision of the MMR vaccination will reduce the number of office visits required. Also, by vaccinating at this age, school officials can identify and track children with incomplete immunisations.
MMR can be given to children as young as 6 months of age, at high risk of exposure, such as during international travel or a community outbreak. However, (according to the ACIP), doses given before 12 months of age cannot be counted toward the two-dose series for MMR.
These children will need two more doses of the vaccine, at 12 months and 4 to 6 years of age.
In the United Kingdom, the first dose is given at 12 months and the second at around 3 years, 4 months.
Most European Union countries immunise at 12 months, with the second dose between 4 and 12 years.
Only Liechtenstein uses the nine to 12-month protocol and recommends that if the first dose of MMR is given between six and eight months of age, three doses of MMR are necessary to complete the immunisation cycle.
Austria recommends only two doses at 10 months and 12months. If the first dose is given in the first year of life, the recommended interval between the doses is three months (minimal interval four weeks).
If the first dose of MMR is given after the first birthday, the second dose is recommended as early as possible with a minimal interval of four weeks.
Malaysia is among the few countries which vaccinate infants with MMR at nine months, with the second dose at 12 months. In Indonesia, the measles vaccine is given at nine months, 24 months, and 7 years, and from 2004, MR at nine months, 18 to 24 months, and seven years).
Measles is a highly infectious disease caused by the RNA paramyxovirus. It was first described by 9th century Persian physician Abu Bekr Rhazes as Hasbah, which means ‘eruption’ in Arabic.
Prior to the vaccine, globally, there were approximately 30 million cases, with two million deaths each year. In many countries, it ranks among the top ten leading causes of toddler deaths and can precipitate protein-calorie malnutrition in childhood.
In 1954, a measles outbreak at a boarding school outside Boston prompted the Boston Children’s Hospital to isolate the virus from throat swabs and blood samples of infected students. The samples that Dr Thomas Peebles obtained from David Edmonston, a 11-year-old schoolboy, led to the successful culture of the measles virus.
Peebles’s superior, Dr John Franklin Enders, often called the ‘Father of Modern Vaccines’, developed the measles vaccine from the Edmonston-B strain, named after David, and used as the basis for most live-attenuated vaccines to this day.
Enders’s team tested the measles vaccine on small groups of children from 1958 to 1960, before beginning trials on thousands of children in New York City and Nigeria. In 1961, it was hailed as 100 per cent effective. The first measles vaccine was licensed for public use in 1963.
Originally, the vaccine was a single dose against measles only, administered at nine months as an injection in the baby’s upper arm. The combined vaccines MMR (MMRII, Merck) was licensed in 1971 and MMRV with chickenpox in 2005.
In May 1974, the World Health Organization (WHO) created the Expanded Program on Immunization (EPI) to increase vaccine coverage among children worldwide.
By 1980, all United Nations member states had their national EPI with varying schedules as determined by their own governments.
In 1984, the WHO EPI created the first standardised vaccination schedule, recommending the following four vaccines against six diseases:
- Tuberculosis (BCG): At birth.
- Diphtheria, Tetanus, and Pertussis (DPT vaccine): At six, 10, and 14 weeks
- Poliomyelitis: At six, 10, and 14 weeks
- Measles: At nine months
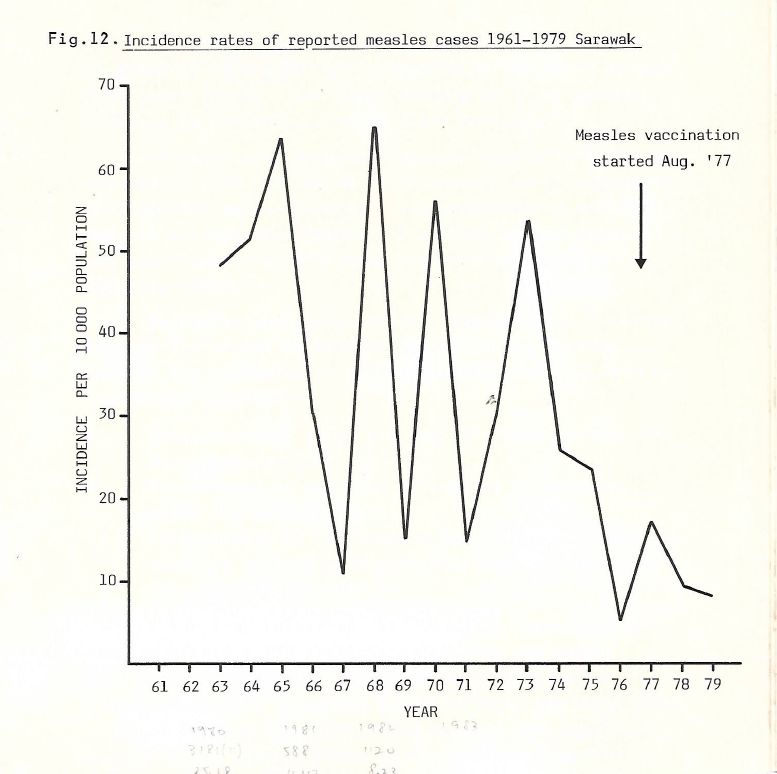
Measles vaccination was started in Sarawak in August 1977, and Peninsular Malaysia in 1982.
It was first given in two doses, the first dose at six months and the second at 12 months. This was changed to a single dose at nine months in September 1980.
In Sarawak, between 1961 and 1977, measles epidemics occurred every two to three years, with peaks of 50 to 60 cases per 10,000 population. After the introduction of the vaccine, the annual incidence dropped to below 10 cases per 10,000 population by 1979.
The provision of Maternal and Child Health (MCH) services in Sarawak began in 1962 (prior to the formation of Malaysia) by local authorities who were responsible for the building, staffing, and training of midwives. On January 1, 1979, the MCH staff and related services were integrated and taken over by the Ministry of Health (MOH).
In Malaysia, from 1982 until 2002, a single dose of measles vaccine was given at nine months.
As a result, the incidence rate of measles dropped from 65.2 cases per 100,000 population in 1982 to between 1.51 and 5.87 cases per 100,000 population between 1989 and 1998.
The timing was changed to 12 months on July 1, 2002, as it was noted that 20 per cent of children who received the measles vaccine at nine months did not develop sufficient antibodies to protect them. More older children were getting measles. The single-dose measles vaccine was replaced by MMR in 2002.
Children in Peninsular Malaysia and Sarawak affected by this change will receive their MMR vaccine when they are a year old.
Those in Sabah will get a single dose of measles when they are six months old because the disease pattern there is different. They will still get MMR at one year. All children will get a booster dose when they are seven years old.
In 2011, Malaysia saw a resurgence of measles. The number of cases jumped from 195 cases in 2013 to 1,934 cases in 2018. About 68.7 per cent of the cases in 2013 were among non-immunised children, compared to 75.9 per cent in 2018.
From April 1, 2016, in response to the 2011 outbreak, and in accordance with the WHO’s 2009 recommendation for countries with high incidence of measles, the MOH changed the MMR schedule to give the first dose at 9 months and second at 12 months.
The second dose MMR scheduled for the first year of primary school was to be discontinued in 2022 when the cohort affected by this change reached 7 years of age. The majority of studies suggests that peak vulnerability will occur between four to nine months of age.
In 2018, there were six measles deaths in Malaysia (all not immunised), five diphtheria deaths (four not immunised), and 22 deaths from whooping cough (19 non-immunised).
Health director-general Dr Noor Hisham Abdullah said that certain individuals who had objected to vaccines had influenced other parents to join them through social media. “Their irresponsible action posed a challenge to the ministry’s efforts, and as a result, infectious diseases that could be prevented with vaccines had increased,” he said.
The WHO has identified vaccine rejection as one of the global health threats for 2019. Despite the availability of a safe and effective vaccine, more than 140,000 worldwide died from measles in 2018, mostly children under the age of 5 years.
For every 1,000 reported measles cases in the United States, one case of encephalitis will result, and two to three deaths.
The incidence by age peaks among children from six months to 4 years of age, with 10 per cent affecting infants below 1 year old.
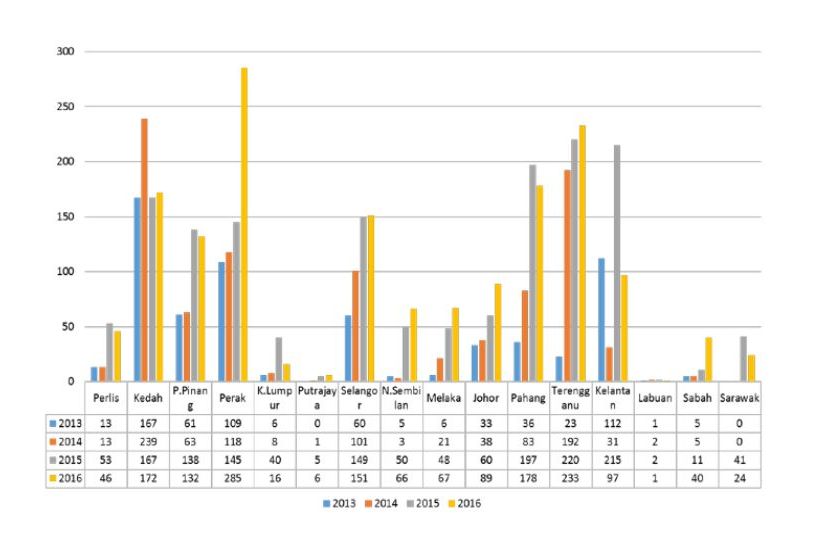
Number of Childhood Vaccine Refusal by States 2013-1016 Malaysia
Does the vaccine work? After two doses of MMR vaccine, about 99 of people out of 100 will be protected against measles, 88 out of 100 will be protected against mumps, and almost everyone will be protected against rubella.
Measles outbreaks among populations which have received two doses of measles-containing vaccine are uncommon.
Both serologic and epidemiologic evidence indicate that measles-containing vaccines induce long lasting immunity in most persons, which is approximately 95 per cent of vaccinated persons examined 11 years after initial vaccination and 15 years after the second dose of MMR.
Should we be giving our children who were vaccinated at nine months and 12 months the third dose of MMR at 7 years? The ACIP and some countries recommend it, since they regard the first dose given below nine months as not counted toward the two-dose series.
Studies indicate better antibody responses in children given at 12 months or older compared with responses in younger children but in children 9 months or older, seroconversion rates still exceed 90 per cent.
In my own practice, I give MMR at 12 months and 4 to 5 years. Parents are asked to bring the infant earlier if there is a measles outbreak or close contact.
Post-exposure prophylaxis with MMR vaccine appears to be effective if the vaccine is administered within three days of exposure.
Instead of giving the third MMR dose to every 7-year-old children who have had two doses (at nine months and 12 months), we should monitor the incidence of measles, mumps, and rubella cases closely, and give third doses as post-exposure prophylaxis during outbreaks or history of exposure.
Within the next few years, we should know if a third dose is indeed needed for most Malaysian children, or whether we should reschedule the two doses to 12 months and above.
The triple jab MMR was banned in Japan in 1993 after some of the 1.8 million children who had been given two types of MMR developed non-viral meningitis and other adverse reactions.
There were three deaths, while eight children were left with permanent handicaps, ranging from damaged hearing and blindness to loss of control of limbs.
Japan manufactures single-dose measles, rubella. and mumps vaccines, and combined measles and rubella vaccine for their vaccination programme.
The Serum Institute of India have stopped manufacturing single rubella vaccines. This mirrors what Sanofi Pasteur did with Rudivax (Rubella vaccine) in 2012.
When the last batch produced expired at the end of January 2015, the single rubella vaccine will no longer be available. The single mumps vaccine has not been available since 2010.
This means that only the single measles vaccine, the measles and rubella combined vaccine, and the MMR vaccine are currently available in India.
Dr Tan Poh Tin is a paediatrician and public health specialist.
- This is the personal opinion of the writer or publication and does not necessarily represent the views of CodeBlue.