KUALA LUMPUR, July 30 — The Health Ministry pledged to criminally charge drug wholesalers and private hospitals, clinics or pharmacies that sell prescription medicines exceeding price ceilings under impending regulations.
Even though drug price controls are supposedly not decided yet since the Health Ministry is still discussing with industry stakeholders, the ministry triggered the policy-making process by starting an online public consultation on the Unified Public Consultation (UPC) portal of the Malaysia Productivity Corporation (MPC), an agency under the Ministry of International Trade and Industry.
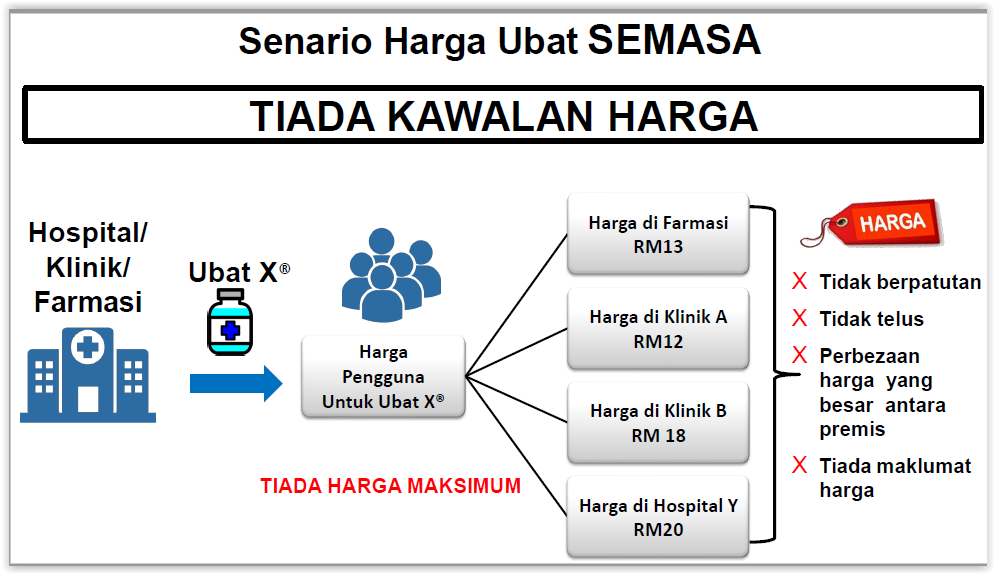
The Health Ministry said Malaysia had no legislation to protect consumers from “unreasonable” medicine prices. But the ministry did not explain how exactly it would determine so-called profiteering, given that the cost of developing a drug is often complex and unknown, and various health care providers claim different costs in their business.
“In line with that, the Ministry of Health (MOH) has taken the initiative to control the price of medicines in Malaysia by using the Price Control and Anti-Profiteering Act 2011 (Act 723),” said the Health Ministry’s Pharmaceutical Services Programme on MPC’s public consultation portal.
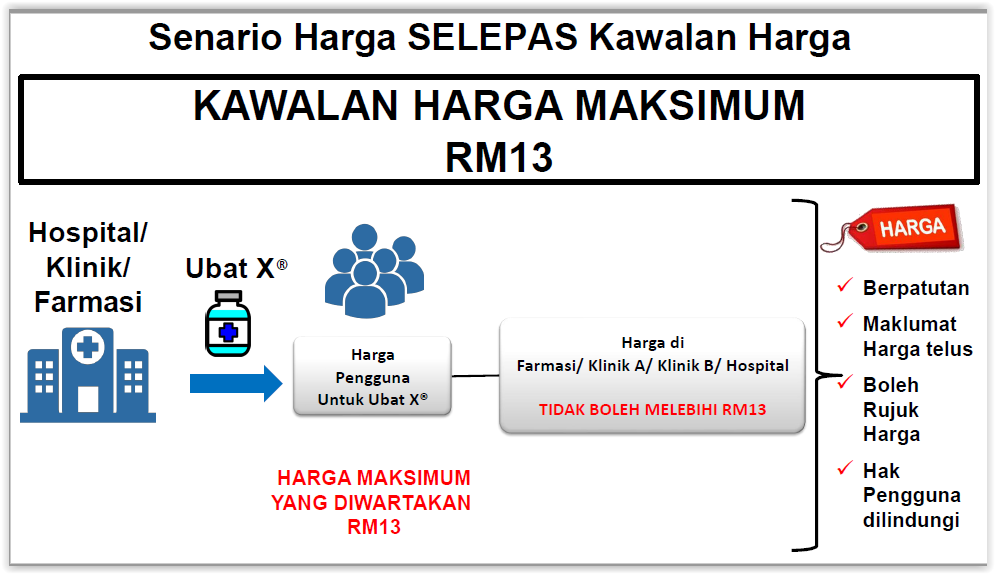
“The following is the drug price control mechanism that will be implemented: maximum wholesale price and maximum retail price. Action will be taken under the law against the sale of medicine that exceeds the maximum price.”
Under the Price Control and Anti-Profiteering Act, profiteering, and selling and buying goods outside the regulated price is punishable with a maximum RM500,000 fine for companies, and for individuals, a maximum RM100,000 fine and three years’ jail. It’s unknown if patients will be prosecuted under the law for purchasing medicines at unregulated prices if drug price controls come into effect.
The Health Ministry’s public consultation on regulating drug prices was opened on MPC’s portal from July 26 to August 12. The consultation is listed under the “Forum” stage, the first of three stages before a draft regulation is finalised.
Putrajaya’s first phase of medicine price controls will target originator drugs, or single-source products, among controlled medicines, which are prescription drugs and medicines dispensed by a pharmacy.
According to an explanatory document by the Health Ministry, both the maximum wholesale and retail prices for drugs will be gazetted and displayed on the ministry’s public domain.
Clinics, pharmacies, and hospitals are required to publicly display the gazetted prices. Consumers will be able to file complaints if medicines are sold above the maximum gazetted prices, which is considered a criminal offence. Regulation of drug prices will be enforced by the Health Ministry’s pharmacy officials appointed by the Ministry of Domestic Trade and Consumer Affairs.
The Health Ministry will use external reference pricing to determine maximum wholesale prices for medicines in Malaysia by looking at the prices of drugs sold in other countries. Maximum retail prices for medicines sold by private pharmacies, hospitals, medical and dental clinics, and haemodialysis centres among others, will then be set through regressive mark-ups on the maximum wholesale price.
But the Health Ministry did not state on MPC’s consultation portal which countries would be referenced or how they would be chosen, how the external reference prices and mark-ups would be calculated, alternative methodologies, which medicines exactly would be targeted, or how drug price controls would impact the entire health care industry.
Expenditure on medicines comprised only 10 per cent of out-of-pocket spending, according to the Health Ministry. Out-of-pocket spending took up 38 per cent of Malaysia’s total health expenditure in 2015.
A 2004 study by Dr Nik Rosnah Wan Abdullah from Universiti Malaya showed that medicine made up just 15 per cent of the bill in private hospitals, while services ranged from 15 per cent to 28 per cent.
Health Ministry’s Justification For Price Controls
In another document, the Health Ministry claimed that drug prices were rising and that medicines were too expensive in the private sector in Malaysia because of a lack of price controls. But the ministry simply showed that spending on medicine in Malaysia, an ageing society, rose 23 per cent from RM3.8 billion in 2011 to RM4.7 billion in 2014.
The Health Ministry also alleged that profiteering could happen at each stage of the drug supply chain (manufacturer/ importer, wholesaler/ distributor, retail/ health care provider), but did not provide any evidence. The ministry merely cited an unreferenced 2012 study that claimed the average sales of a community pharmacy in Penang were between 30.3 per cent and 148.28 per cent higher than in Australia.
The Health Ministry justified drug price controls by saying that only 30 per cent of pharmaceutical companies voluntarily displayed their product prices after it issued the “Good Pharmaceutical Trade Practice” guidelines to the industry in 2015 to promote fair and transparent pricing.
The Health Ministry acknowledged that some studies show regulating medicine prices may delay the entry of new drugs into the market, especially for low-income economies. But it cited an unreferenced 2009 study that claimed European countries still led in drug innovation, despite using drug price controls, compared to the United States’ free market.
Most pharmaceutical companies’ research and development (R&D) spending, the Health Ministry alleged, went to low-risk programmes that only had minor clinical benefits over existing treatments.
“In line with that, access of new and innovative drugs entering the Malaysian market and the growth of the pharmaceutical industry will be given due attention later during the implementation of drug price controls.
“In fact, the industry can benefit from this initiative through transparent price information that will enable more effective business planning,” said the Health Ministry.
Drug Price Controls Will Kill Private Hospitals
Association of Private Hospitals of Malaysia (APHM) president Dr Kuljit Singh, however, yesterday publicly appealed to Health Minister Dzulkefly Ahmad to review his decision to regulate drug prices.
Dr Kuljit pointed out that medical tourists from neighbouring countries have been moving to Malaysia’s private hospitals in recent years, indicating the affordability and quality of medical care here.
The Asean Post reported last May that the number of medical tourists coming to Malaysia surged to 1.2 million arrivals last year from 643,000 in 2011. Revenue generated in that period rose from US$127 million (RM523 million) in 2011 to US$362 million (RM1.5 billion) in 2018.
“We are trying our best to provide treatment to our rakyat at a cost which is one of the cheapest in the region,” Dr Kuljit said in his opening speech at the 27th APHM International Healthcare Conference and Exhibition here yesterday.
“Here, I would like to ask here if there are indeed moves to regulate pricing in other service industries, like hotels and restaurants as well? If the answer is no, then why target just private hospitals?”
Dr Kuljit Singh, APHM president
Dr Kuljit told CodeBlue later that drugs only made a small percentage of the bill to patients in private hospitals, claiming that medicine price regulations would not cut health care costs.
“Just by controlling drug prices, they’ll kill private hospitals’ business,” he said.
Industry Caught By Surprise
An industry source said that the industry was taken aback by the Health Ministry starting a public consultation on MPC’s portal about regulating medicine prices, as if it were running a regulatory impact assessment (RIA).
“We were told that this process of RIA will happen much later, not now,” the source told CodeBlue on condition of anonymity due to the sensitivity of negotiations.
“This seems to be a bit premature in the process because it’s still at the conceptual level.”
At the time of writing, five comments were posted on the Health Ministry’s online consultation, all of which were made by anonymous authors. One hidden comment was marked “abusive”.
The industry source noted that previous RIAs run by other ministries and coordinated by the MPC were more thorough, unlike the Health Ministry’s that lacked responses from the industry, business chambers, patient groups, and civil society.
“Now, it sounds like Twitter,” the industry source complained. “Is this just a tick box exercise, or is this meaningful?”
The source also said although all health stakeholders have been engaging with the Health Ministry about drug price controls, none have discussed this issue with other ministries yet.
“I think everyone was expecting that to happen, but suddenly there’s a public consultation. It looks like it’s already decided.”








