KUALA LUMPUR, June 17 — Malaysia is looking to shorten the 12-week dosage interval for AstraZeneca-Oxford’s Covid-19 vaccine, though a decision depends on the steady delivery of the shot.
Vaccine Minister Khairy Jamaluddin yesterday said there is a possibility of expediting the second vaccine dose for full protection amid the spread of more contagious variants in the country, though he did not specify the dosing interval.
“We are thinking of shortening the AstraZeneca interval. The only problem I have here is with the vaccine supply. A part of the AstraZeneca supply from COVAX was delayed and there was also a slight setback on the supply we ordered from Thailand.
“We are recalculating our delivery schedule right now to see whether we can shorten the interval period for AstraZeneca,” Khairy said during The Oxford and Cambridge Society Malaysia’s “The Path to Herd Immunity” discourse yesterday.
Malaysia has directly purchased 6.4 million doses of the two-dose AstraZeneca vaccine, manufactured by Thailand’s Siam Bioscience, besides ordering another 6.4 million Covid-19 vaccine doses from COVAX, a global vaccine sharing programme backed by the United Nations. COVAX’s AstraZeneca vaccine delivery to Malaysia is manufactured by South Korea’s SK Bioscience.
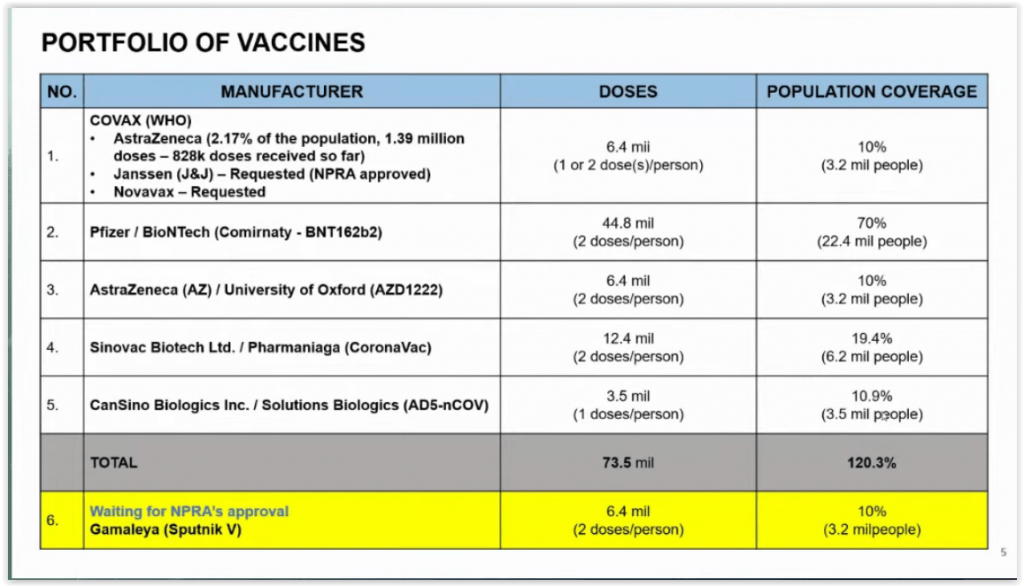
COVAX has delivered 828,000 out of 1.39 million AstraZeneca vaccine doses to Malaysia so far. Besides AstraZeneca’s shot, the government has also requested coronavirus vaccines by US vaccine developers Johnson & Johnson and Novavax from COVAX for Malaysia’s total order of 6.4 million doses. Local pharmaceutical regulators have approved Johnson & Johnson’s single-shot Janssen vaccine for use in Malaysia.
The recommended interval for the AstraZeneca vaccine is eight to 12 weeks. Canada’s Ontario province recently moved to shorten the interval between AstraZeneca vaccine doses to eight weeks amid concerns about a rise in cases of the new Delta variant in Ontario, local media reported. The UK has also expedited the second Covid-19 vaccine dose from 12 to eight weeks for vulnerable groups to tackle increasing cases of the Delta variant first found in India.
Recently published real-world data by Public Health England shows that two doses of AstraZeneca-Oxford’s Covid-19 vaccine is 92 per cent effective against hospitalisation caused by the Delta variant and prevents death.
This means a fully vaccinated person has a 92 per cent lower risk of developing severe illness from the Delta variant compared to those who have not been vaccinated.
Khairy said the government is also mulling heterologous Covid-19 vaccination using AstraZeneca as the first dose and Pfizer-BioNTech as the second, following studies which show that a mix and match of the two vaccines can boost efficacy against different Covid-19 variants.
“We don’t want to make a quick decision on this before getting more data. The data points are coming in for heterologous vaccinations. Once the technical working group is clear on that, I’ll advise the committee that I chair with the Health Minister and we will implement heterologous vaccinations. It is possible that we will end up doing this,” Khairy said.
“Many countries are doing this to boost the neutralising antibodies, boost the efficacy, and when you are facing vaccine supply constraints, you can mix things up and ensure that the effective vaccines are still there.”