As the 194 members states of the World Health Organisation re-convenes next week for the 73rd World Health Assembly, a global coalition of 29 think tanks from 19 countries, including the Galen Centre for Health & Social Policy, has called on governments around the world to implement reforms which accelerate access to medicine, particularly those crucial to the COVID-19 response.
These recommended reforms address two specific issues, namely the cost and access to medicines.
The need to tackle escalating high out-of-pocket costs related to healthcare, has grown increasingly urgent in light of the global coronavirus pandemic. Health complications which include escalation in preventable non-communicable diseases such as cancer, cardiovascular disease and diabetes, have been exacerbated by lock-downs and measures intended to restrict movement and social interactions.
In many developing countries, communities, households and individuals are meeting existing and new healthcare costs through out of pocket expenses, a significant portion of which involve the purchase of medicines.
To improve access to medicines, unnecessary medicine price inflators should be removed. Many of these can be traced back to import tariffs imposed by governments.
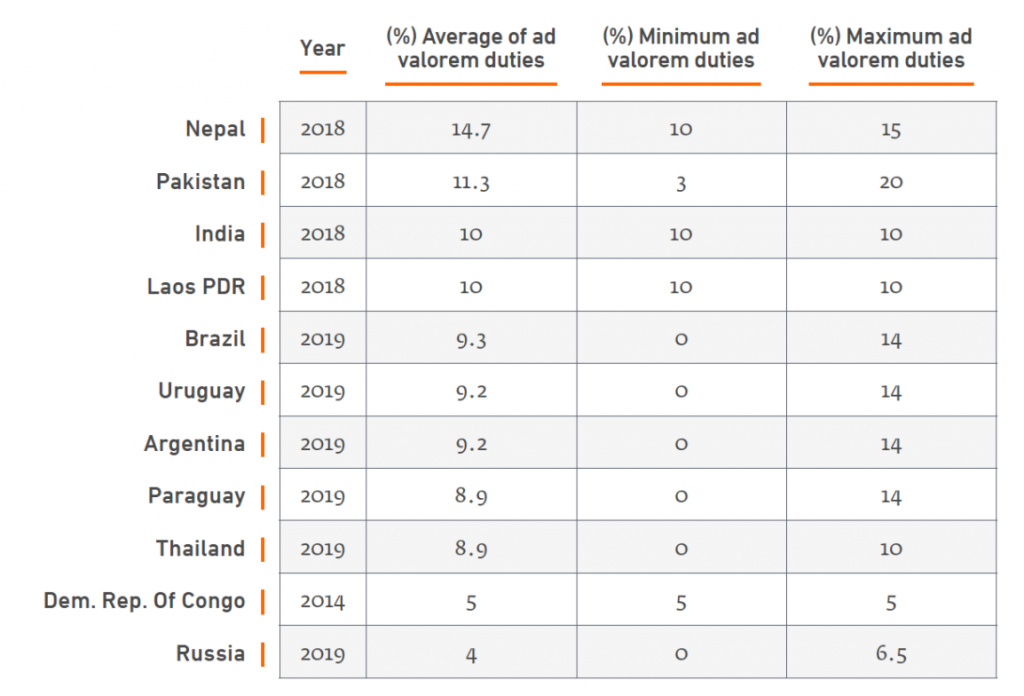
India, Nepal and Pakistan have the world’s top three tariff rates on medicines. Pakistan, which has the highest, imposes a 20 percent tariff. Even vaccines which are considered essential and life-saving for many populations facing vaccine preventable diseases are not spared. India imposes a rate of 10 percent. Patients end up paying for medicines whose prices could be lower
Consider that by abolishing these tariffs alone, patients would save up to US$6.2bn in China, US$737m in India, and US$252 in Indonesia.
Eliminating domestic taxes such as value-added taxes (VAT) or goods and services taxes (GST) as well as excise and stamp duties would make also make a major difference. These taxes could sometimes make up 20 – 30 percent of the price that people pay for medicines.
Accelerating access to medicines, especially those classified as essential, has become more urgent than ever before.
The availability of new medicines is often affected by delays and bottlenecks involved in the regulatory process at the national level. Patients are unable to access innovative treatments even after they have already been declared safe and efficacious by a stringent regulatory authority such as the European Medicines Agency (EMA). It can take many years after the global launch for new medicines to reach patients.
Together with India, Taiwan, Singapore and South Korea, Malaysia was named as one of the countries where delays averaged between 400-500 days. Patients in Indonesia wait for almost three years.
The decision for new treatments to be included into national drug formularies can also be a factor to accelerate or delay a medicines availability in the public health system. Improving the efficiency of the decision-making process involved has the potential to improve reimbursements and most importantly, patient access to life-saving medicines.
This is going to be a major hurdle needing to be addressed with the distribution and use of COVID-19 treatments that are currently under development.
Biopharmaceutical innovation is at the frontlines of developing life-saving COVID-19 treatments, especially in the area of a preventive vaccine. Key to that is ensuring the integrity of intellectual property (IP) rights which facilitate cooperation and the sharing of proprietary data, and technology between competing companies, organisations and countries.
We know that the IP system is working because there is now over 1240 active clinical trials on 534 unique therapies, and 43 unique vaccines being tested. There is a global framework in place which allows for resources to be shared with stakeholders in the private sector with the aim of quickly manufacturing billions of doses and distribute them globally.
These reforms are low-cost practical measures which governments can immediately take to rapidly improve and accelerate access to quality medicines and progressing towards the right to health. They should also be taken as part of addressing the COVID-19 outbreak in their borders.
As new treatments and vaccines for COVID-19 become available, by implementing reforms on the regulatory and trade barriers now, countries can help prepare the ground for the medicines to be made available as quickly as possible for their citizens.








